Received: Mon 17, Mar 2025
Accepted: Fri 04, Apr 2025
Abstract
Translational research and clinical trials on malignant manifestations utilizing low temperatures in biomedical science and practice have provided significant momentum to fundamental studies and clinical research on malignancies. A retrospective survey of 8,371 cancer patients who underwent 10,892 cryoablation treatments was conducted. Among them, 25 patients met the following criteria: advanced cancer stage (III or IV), a life expectancy of <1 year, exclusive treatment with cryoablation, and survival of >10 years. The longest survival period recorded was 18 years, with nine patients (36%) surviving >16 years, and 18 patients (72%) still alive at follow-up. Based on treatment history, four long-term survival patterns post-cryoablation were identified: i) long-term remission following a single freezing session; ii) long-term remission after multiple freezing treatments within the first 1-2 years; iii) long-term remission for several years post-freezing, followed by relapse after 8-10 years, requiring repeated freezing, leading to improvements and further recurrences; and iv) sustained remission through continuous cryoablation, enabling long-term "living with cancer." These findings suggest that cryoablation effectively extends survival in patients with advanced cancer, potentially due to the abscopal effect of cryoimmunology.
Keywords
Cancer, cryosurgery, tumor cryoablation, cryoimmunology, abscopal effect
1. Introduction
The primary goal of cancer treatment is to prolong patient survival. Conventional treatments such as chemotherapy and radiotherapy often fail to achieve long-term survival in patients with unresectable advanced cancers. Despite technological advancements in imaging and minimally invasive tumor ablation, improving patient survival remains a significant challenge [1, 2]. Translational research and clinical trials on malignant manifestations, employing low temperatures in biomedical science and practice, have significantly advanced fundamental studies and clinical research on malignancies. This progress has benefited academic and clinical researchers, physicians, scientists, and leading brand exhibitors worldwide [3, 4].
Specific cell debris, the cryocell detritus or cryocell debris (CCD), was discovered and observed in vivo in early 2000s by Korpan, using a cryosurgical approach [5, 6]. The vascular capillary changes and circulatory stagnation demonstrate the anti-angiogenesis mechanism, which, together with cryoaponecrosis and cryoapoptosis, are some of the main mechanisms of biological tissue injury following the low temperature exposure. This presented study retrospectively analyzes patients with advanced-stage cancer who exclusively received cryoablation and survived for >10 years, aiming to evaluate the effectiveness of this local treatment in improving long-term survival.
2. Materials and Methods
Between January 2003 and July 26, 2023, 8,371 cancer patients underwent 10,892 cryoablation procedures at Fuda Cancer Hospital (Guangzhou, China) and the International Institute of Cryosurgery (Rudolfinerhaus Clinic, Vienna, Austria) as well as the Department of General Surgery №1, Bohomolets National Medical University (Kyiv, Ukraine). A retrospective survey identified patients who met the following inclusion criteria:
i) Advanced tumors (stage III or IV) and a life expectancy of <1 year.
ii) Tumor recurrence after surgery or presence of unresectable tumors.
iii) Failure of conventional treatments, including chemotherapy and radiotherapy.
iv) Exclusive treatment with cryoablation post-admission.
v) Survival >10 years post-cryotherapy.
Cryoablation was performed using an argon-helium cryosystem (Endocare or Cryo-Hit, USA) and liquid nitrogen (FreezeForce1, Austria), either percutaneously under ultrasound and/or CT guidance [7] or via open cryosurgery [5, 8, 9]. Tumor response was assessed through CT and MRI scans before the initial cryoablation and 3 months post-treatment. Tumor size changes were categorized according to the response evaluation criteria for substantial tumors as CR, PR, SD, or progressive disease. Survival duration was calculated from the date of cryoablation to the date of death or last follow-up.
3. Results
Of the 8,371 patients who underwent cryoablation, 25 met the study criteria (Table 1). These cases included hepatocellular carcinoma (HCC), hepatic colorectal metastases (HCRM), non-small cell lung cancer (NSCLC), breast cancer, and ovarian cancer. Tumor response was classified as complete response (CR) in 15 patients (60%), partial response (PR) in seven patients (28%), and stable disease (SD) in three patients (12%). The longest survival recorded was 18 years, with nine patients (36%) surviving >16 years and 18 patients (72%) still alive at follow-up.
TABLE 1: Follow-up
results after cryoablation in 25 patients with advanced cancer.
|
Cancer Type |
Cases |
CR (n) |
PR (n) |
SD (n) |
Survival (10-15 years) |
Survival (≥16 years) |
Still alive |
|
HCC |
7 |
4 |
3 |
0 |
5 |
2 |
6 |
|
HCRM |
4 |
3 |
1 |
0 |
3 |
1 |
3 |
|
NSCLC |
9 |
4 |
2 |
3 |
6 |
3 |
5 |
|
Breast cancer |
4 |
3 |
1 |
0 |
3 |
1 |
3 |
|
Ovarian cancer |
1 |
1 |
0 |
|
0 |
0 |
1 |
|
Total |
25 |
15 |
7 |
3 |
17 (68%) |
7 (28%) |
18 (72%) |
HCC: Hepatocellular Carcinoma; HCRM: Hepatic Colorectal Metastases; NSCLC: Non-Small Cell Lung Cancer; CR: Complete Response; PR: Partial Response; SD: Stable Disease.
Based on patient treatment histories, post-cryoablation survival patterns can be categorized into four distinct types:
Type I: Long-term remission after a single cryoablation session. In these rare cases (n=3), the patient underwent cryoablation as the sole treatment. Following ablation, the lesion disappeared, leading to long-term disease-free survival (Figure 1).
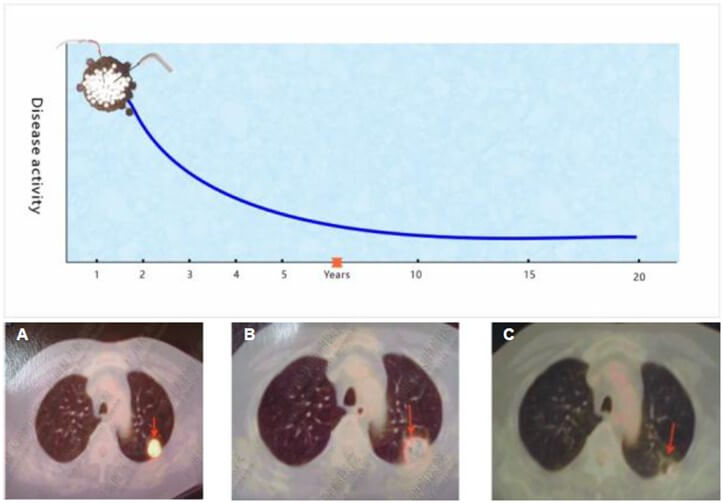
The right image displays a tumor in the posterior segment of the left lung. The middle image shows tumor expansion post-freezing with a reduced CT value, indicating successful ablation. The left image confirms no recurrence during an 18-year follow-up.
Type II: Long-term remission after repeated cryoablation. Initially, these patients (n=13) experienced short-term remission after cryoablation, followed by relapses. Subsequent re-freezing led to multiple remissions, eventually resulting in a prolonged progression-free state, allowing them to "live with cancer" for many years (Figure 2).
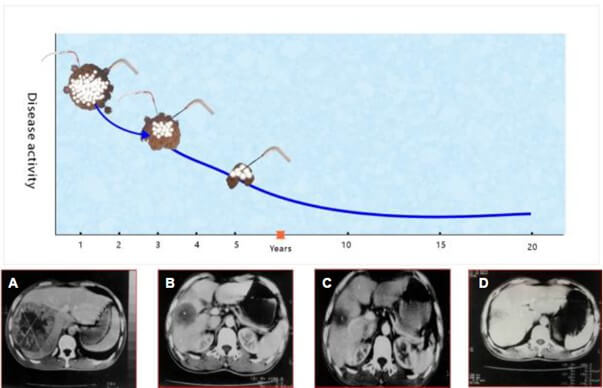
From right to left: A) Tumor necrosis following cryoablation of a large tumor in the right hepatic lobe; B & C) Tumor shrinkage, persistence, and subsequent re-freezing; D) A follow-up CT scan 17 years later showing tumor disappearance.
Type III: Multiple relapses after prolonged remission. Following an extended remission period post-cryoablation, these patients (n=9) typically experienced tumor relapse after 8-10 years. The treatment cycle included cryoablation, remission, relapse, and re-freezing (Figure 3). Most patients in this category did not survive the follow-up period.
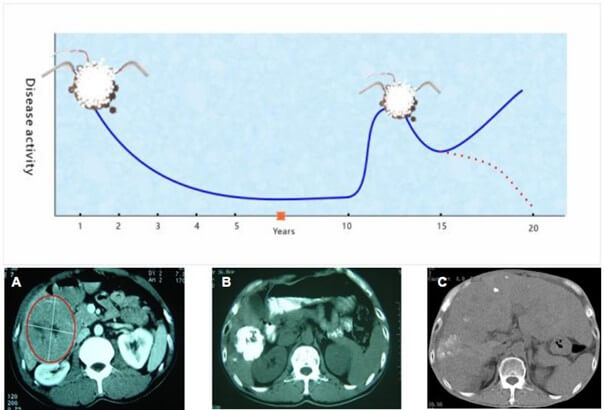
A) A large tumor in the right hepatic lobe before cryoablation. B) Significant tumor shrinkage two months post-cryoablation. C) No recurrence of the original intrahepatic tumor after 10 years.
Type IV: Persistent tumor recurrence with repeated cryoablation. In this pattern (n=3), patients experienced several months of remission following each cryoablation session before new tumor masses emerged. These recurrent tumors were treated with additional cryoablation, leading to a sustained "living with cancer" status (Figure 4).
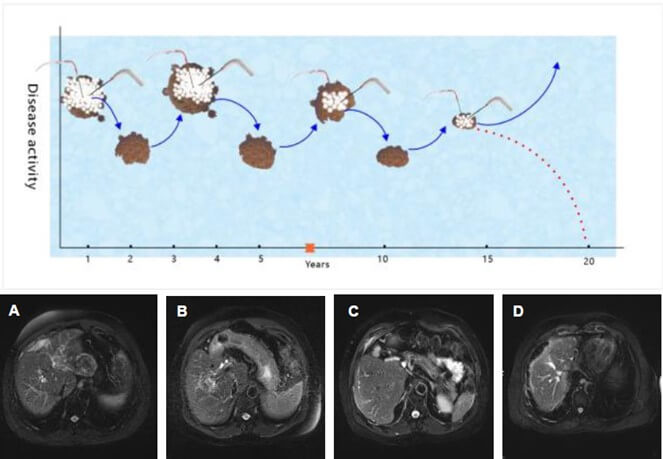
Over the years, new tumors developed in the liver, requiring multiple cryoablation treatments and resulting in long-term disease management.
4. Discussion
Extending the survival of patients with advanced cancer remains a significant challenge. Cryoablation, a minimally invasive technique, has shown promise as a safe and effective treatment for malignancies [10], leading to tumor regression and, in some cases, complete disappearance [11]. Experimental evidence suggests that cryoablation induces a cryoimmunological response similar to autoimmune reactions against altered self-antigens. The immune-stimulatory effects of freezing can act as an adjuvant when combined with specific antigens. Cryo-modified protein components, formed at ultralow temperatures, play a crucial role in generating immunological responses [12, 13].
In this cohort of 25 patients with advanced cancer, 60% exhibited a complete tumor response, while 28% achieved a partial response. However, the primary goal of cancer treatment extends beyond tumor response- it aims to improve long-term survival.
Remarkably, all 25 patients survived for more than 10 years following cryoablation, with 64% living between 10 and 15 years and 36% surviving for ≥16 years. Currently, 72% of these patients remain alive. Although this sample represents a small subset of the 8,371 patients who underwent cryoablation, it is notable that these patients had advanced-stage cancer with an expected survival of no more than one year. Importantly, they received cryoablation as their sole treatment, without any other "anti-cancer" therapies, yet experienced significantly prolonged survival. This finding strongly suggests that cryoablation may substantially extend patient survival.
Metastasis is the leading cause of cancer-related mortality. Preventing or eliminating metastases is crucial for long-term survival. Clinical studies have reported the disappearance of distant metastases following cryoablation of the primary tumor [14]. Likewise, experimental research has demonstrated a significant reduction in metastases in cryoablation-treated groups compared to controls [15], a phenomenon known as the abscopal effect.
Cryoablation also modulates the tumor microenvironment, converting an immunologically “cold” tumor into a “hot” one (ICIE). Experimental studies have shown that cryoablation of breast cancer leads to a >100-fold increase in the ratio of CD8+ cytotoxic T cells to immunosuppressive regulatory T cells, both within primary tumors and in untreated distant tumors [16]. This transformation is initiated by tumor cell necrosis surrounding the cryoprobe, leading to the release of intracellular organelles, antigens, and damage-associated molecular patterns (DAMPs), such as DNA and heat shock proteins. Upon encountering these antigenic substances, dendritic cells become activated, phagocytize DAMPs, and initiate an immune response via nuclear factor kappa-light-chain-enhancer signaling. This process involves CD80/86 co-stimulation, antigen presentation via major histocompatibility complex molecules, and T cell receptor activation, ultimately triggering a systemic immune response [1].
The therapeutic impact of cryoablation in reducing cancer metastasis is largely dependent on CD4+ T cells, which play a crucial role in generating long-lasting memory anti-tumor immunity [17]. Cryoablation-induced tumor microenvironment remodeling also leads to an expansion of tumor-specific T cells, as evidenced by increased tumor-infiltrating lymphocytes (TILs). Studies have reported elevated TIL levels in multiple cancers, including lung, colon, ovarian, and breast cancers [18, 19], particularly at the tumor stroma and invasive margins [19]. Notably, specific TIL responses have also been detected in distant untreated metastatic tumors following primary tumor cryoablation.
Unlike thermal ablation techniques, which denature tumor antigens, cryoablation preserves potential tumor-associated antigens (TAAs) by rapidly freezing tumor tissues. This preservation enhances the immune response. For example, Ravindranath et al. [20] found a significant increase in serum gangliosides exclusively following cryoablation, but not after radiofrequency ablation or surgical resection, in patients with colorectal cancer liver metastases. Additionally, antiganglioside IgM titers increased only after freezing. Another key advantage of cryoablation is the immediate post-thaw rapid reperfusion of blood flow, which facilitates immune cell infiltration into the tumor- an effect not observed with thermal ablation, where blood flow stagnation occurs during heating [16].
Moreover, cryoablation is associated with lower recurrence and metastasis rates compared to surgical resection. Khan et al. [21] demonstrated in an experimental breast cancer model that no recurrence or metastasis occurred after cryoablation, whereas 40% of mice in the resection group developed local recurrence and lung metastases. Clinical observations further support the survival benefits of cryoablation. Mou et al. [22] reported that patients with bladder cancer treated with endoscopic balloon cryoablation had significantly higher survival rates than those treated with transurethral resection of bladder tumors.
Although tumor resection can reduce tumor burden and restore systemic adaptive immunity, it may also activate wound healing programs that induce immunosuppression, primarily via myeloid-derived suppressor cells. This can promote tumorigenic niches and inhibit T cell activity. In contrast, cryoablation increases TIL accumulation, especially in the stroma and invasive margins, while inducing cytotoxic T lymphocyte (CTL) responses in distant untreated tumors [23]. These effects warrant further investigation.
The newly formed cryocoagulated protein components (cryomodified protein components) are crucial in cryoimmunology from the perspective of the formation of immunological substances at ultralow temperatures. Dendritic cells and cryocell detritus (cryocell debris) formed in living biological tissue after exposure to ultralow temperature in vivo may be an indication of one of the essential mechanisms involved in the cryoimmunological response of living structures to the impact of ultralow temperature exposure [13, 24, 25].
5. Conclusion
This study analyzed the long-term survival outcomes of 25 patients with advanced-stage malignancies who survived for more than 10 years following cryoablation therapy. The survival patterns of these patients were categorized into four distinct types:
i) Long-term remission after a single cryoablation session.
ii) Long-term remission following multiple cryoablation treatments.
iii) Multiple relapses after prolonged remission.
iv) Continuous cryoablation with ongoing remission, achieving long-term "living with cancer".
These findings suggest that cryoablation significantly extends survival in patients with advanced malignancies, likely due to the abscopal effect of cryoimmunology, which triggers systemic anti-tumor responses. Further research is needed to clarify the underlying immunological pathways and optimize cryoablation strategies for better long-term outcomes.
Conflicts of Interest
None.
Funding
None.
Data Availability Statement
All relevant datasets generated for this study are within this paper.
Ethics Statement
Written informed consent was obtained from patients with malignant diseases- hepatocellular carcinoma, hepatic colorectal metastases, non-small cell lung cancer, breast cancer, and ovarian cancer- in accordance with the principles of the Declaration of Helsinki, 1964, and its later amendments. The patient signed a declaration of consent for the clinical study that uses cryoablation after detailed explanation with a surgeon-physician about the purpose, manner, and course of events of the planned cryo-treatment, and this is documented in the clinical history of the patient.
Acknowledgments
We thank the scientists and managers at Guangzhou Fuda Cancer Hospital, in partnership with Jinan University School of Medicine (Guangzhou, China), as well as the International Institute of Cryosurgery at Rudolfinerhaus Clinic (Vienna, Austria), for their close research collaboration with the Department of General Surgery No. 1 at Bohomolets National Medical University (Kyiv, Ukraine). Finally, we would also like to thank Prof. Dr. Yaron Har-Shai, past president of the International Society of Cryosurgery, from the Technion-Israel Institute of Technology (Haifa, Israel) and Prof. Dr. Yueyong Xiao, past president of both the International Society of Cryosurgery and the Asian Society of Cryosurgery, Peking University People’s Hospital (Beijing, China), for their academic discussions and expert opinions.
Abbreviations
CCD: Cryocell Detritus or Cryocell Debris
CT: Computer Tomography
CR: Complete Response
PR: Partial Response
SD: Stable Disease
REFERENCES
1. Jibing Chen, Wei Qian, Feng Mu, et
al. “The future of cryoablation: An abscopal effect.” Cryobiology, vol.
97, pp. 1-4, 2020. View at: Publisher
Site
| PubMed
2. Chakradhar Yakkala, Alban Denys 1,
Lana Kandalaft, et al. “Cryoablation and immunotherapy of cancer.” Curr Opin
Biotechnol, vol. 65, pp. 60-64, 2020. View at: Publisher
Site
| PubMed
3. N N Korpan “Hepatic cryosurgery for
liver metastases. Long-term follow-up.” Ann Surg, vol. 225, no. 2, pp.
193-201, 1997. View at: Publisher
Site
| PubMed
4. Nikolai N Korpan “A history of
cryosurgery: its development and future.” J Am Coll Surg, vol. 204, no.
2, pp. 314-324, 2007. View at: Publisher
Site
| PubMed
5. N.N. Korpan “Cryosurgery:
ultrastructural changes in pancreas tissue after low temperature exposure.” Technol
Cancer Res Treat, vol. 6, no. 2, pp. 59-67, 2007. View at: Publisher
Site
| PubMed
6. Nikolai N Korpan “Cryosurgery: early
ultrastructural changes in liver tissue in vivo.” J Surg Res,
vol. 153, no. 1, pp. 54-65, 2009. View at: Publisher
Site
| PubMed
7. Lizhi Niu 1, Liang Zhou, Nikolai N
Korpan, et al. “Experimental study on pulmonary cryoablation in a porcine model
of normal lungs.” Technol Cancer Res Treat, vol. 11, no. 4, pp. 389-394,
2012. View at: Publisher Site | PubMed
8. N.N. Korpan, JV
Zharkov “Cryosurgery
and Cryotechnology – Future Safety in Medicine.” In: Korpan NN, ed. Basics of
Cryosurgery. Vienna: Springer, New York, Vienna, Sydney, Tokyo, Munich, pp.
9-11, 2001.
9. N.N. Korpan “Basic Cryosurgical
Techniques.” In: Korpan NN, ed. Atlas of Cryosurgery. Vienna: Springer, New
York, Vienna, Sydney, Tokyo, Munich, pp. 85-105, 2001.
10. Nicholas Pigg, Robert C Ward
“Cryoablation for the Treatment of Breast Cancer: A Review of the Current
Landscape and Future Possibilities.” Acad Radiol, vol. 30, no. 12, pp.
3086-3100, 2023. View at: Publisher
Site
| PubMed
11. Mitsuko Yodoya, Takao Hiraki,
Toshihiro Iguchi, et al. “Disappearance of Renal Cysts Included in Ice Ball
During Cryoablation of Renal-Cell Carcinoma: A Potential Therapy for
Symptomatic Renal Cysts?” J Vasc Interv Radiol, vol. 28, no. 6, pp.
869-876, 2017. View at: Publisher
Site
| PubMed
12. Martijn H M G M den Brok, Roger P M
Sutmuller, Robbert van der Voort, et al. “In situ tumor ablation creates
an antigen source for the generation of antitumor immunity.” Cancer Res,
vol. 64, no. 11, pp. 4024-4029, 2004. View at: Publisher
Site
| PubMed
13. Nikolai N Korpan, Anatoliy N
Goltsev, Olexii I Dronov, et al. “Bondarovych, Cryoimmunology: Opportunities
and challenges in biomedical science and practice.” Cryobiology, vol.
100, pp. 1-11, 2021. View at: Publisher
Site
| PubMed
14. Aswin V Kumar, Sharla G Patterson,
Michael J Plaza “Abscopal Effect following Cryoablation of Breast Cancer.” J
Vasc Interv Radiol, vol. 30, no. 3, pp. 466-469, 2019. View at: Publisher
Site
| PubMed
15. Tatiana P Grazioso, Nabil Djouder
“The forgotten art of cold therapeutic properties in cancer: A comprehensive
historical guide.” iScience, vol. 26, no. 7, pp. 107010, 2023. View at: Publisher
Site
| PubMed
16. Wenquan Ou, Samantha Stewart, Alisa
White, et al. “In-situ cryo-immune engineering of tumor microenvironment
with cold-responsive nanotechnology for cancer immunotherapy.” Nat Commun,
vol. 14, no. 1, pp. 392, 2023. View at: Publisher
Site
| PubMed
17. Kun He, Ping Liu, Lisa X Xu “The
cryo-thermal therapy eradicated melanoma in mice by eliciting CD4(+)
T-cell-mediated antitumor memory immune response.” Cell Death Dis, vol.
8, no. 3, pp. e2703, 2017. View at: Publisher
Site
| PubMed
18. Wei-Ting Hwang, Sarah F Adams, Emin
Tahirovic, et al. “Prognostic significance of tumor-infiltrating T cells in
ovarian cancer: a meta-analysis.” Gynecol Oncol, vol. 124, no. 2, pp.
192-198, 2012. View at: Publisher
Site
| PubMed
19. Franck Pagès, Amos Kirilovsky,
Bernhard Mlecnik, et al. “In situ cytotoxic and memory T cells predict
outcome in patients with early-stage colorectal cancer.” J Clin Oncol,
vol. 27, no. 35, pp. 5944-5951, 2009. View at: Publisher
Site
| PubMed
20. Mepur H Ravindranath, Thomas F Wood,
Daniel Soh, et al. “Cryosurgical ablation of liver tumors in colon cancer
patients increases the serum total ganglioside level and then selectively
augments antiganglioside IgM.” Cryobiology, vol. 45, no. 1, pp. 10-21,
2002. View at: Publisher Site | PubMed
21. Sonia Y Khan, Michael W Melkus,
Fahmida Rasha, et al. “Tumor-Infiltrating Lymphocytes (TILs) as a Biomarker of
Abscopal Effect of Cryoablation in Breast Cancer: A Pilot Study.” Ann Surg
Oncol, vol. 29, no. 5, pp. 2914-2925, 2022. View at: Publisher
Site
| PubMed
22. Zezhong Mou, Yiling Chen, Zheyu
Zhang, et al. “Cryoablation inhibits the recurrence and progression of bladder
cancer by enhancing tumour-specific immunity.” Clin Transl Med, vol. 13,
no. 5, pp. e1255, 2023. View at: Publisher Site | PubMed
23. Katrina F Chu, Damian E Dupuy
“Thermal ablation of tumours: biological mechanisms and advances in therapy.” Nat Rev Cancer, vol. 14, no.
3, pp. 199-208, 2014.
View at: Publisher
Site
| PubMed
24. Nikolai N
Korpan, Kecheng Xu, Philipp Schwarzinger, et al. „Cryo-Assisted Resection En Bloc, and
Cryoablation In Situ, of Primary Breast Cancer Coupled With
Intraoperative Ultrasound-Guided Tracer Injection: A Preliminary Clinical
Study.” Technol Cancer Res Treat, vol. 17, pp. 1533034617746294, 2018.
View at: Publisher Site | PubMed
25. Lihua He, Lizhi Niu, Nikolai N Korpan, et al. “Clinical Practice Guidelines for Cryosurgery of Pancreatic Cancer: A Consensus Statement From the China Cooperative Group of Cryosurgery on Pancreatic Cancer, International Society of Cryosurgery, and Asian Society of Cryosurgery.” Pancreas, vol. 46, no. 8, pp. 967-972, 2017. View at: Publisher Site | PubMed
