Received: Sat 19, Jul 2025
Accepted: Tue 05, Aug 2025
Abstract
Background: Comprehensive preoperative CT assessment is essential for ventral hernia repair, yet no standardized reporting framework exists. This study evaluated the completeness of preoperative abdominal CT reports and developed an evidence-based protocol to guide standardized reporting for abdominal wall reconstruction (AWR).
Methods: We conducted a systematic evaluation of CT reporting completeness in 834 patients who underwent elective transversus abdominis release (TAR) at the Cleveland Clinic Center for Abdominal Core Health between January 2020 and December 2024. A panel of AWR experts defined 16 CT-based parameters deemed essential for surgical planning, and their clinical relevance was validated through a global survey of 61 AWR surgeons. Radiologic reports were assessed for documentation of these parameters and compared with intraoperative findings and registry data from the Abdominal Core Health Quality Collaborative (ACHQC). Parameters were classified as either generalizable or patient specific. A multi-criteria decision analysis using the analytic hierarchy process was applied to prioritize features for standardized reporting.
Results: Overall documentation completeness was limited, with a median of 34.4%. Although surgeons rated defect width as the most critical parameter for operative planning, it was documented in only 32.6% of CT reports. Patient-specific findings demonstrated higher overall reporting rates (median 87.8%), though key features such as mesh presence and anatomical mesh plane were documented in only 36.2% and 1.7% of applicable cases, respectively. Multi-criteria decision analysis identified defect size, Tanaka index, anatomical hernia location, presence of prior mesh, old mesh plane and concurrent inguinal or stomal site hernia as the most critical parameters for preoperative evaluation.
Conclusion: Substantial gaps exist between CT reporting and the informational needs of AWR surgeons. We propose the "STAMP-C" framework as a pragmatic, consensus-driven model to standardize ventral hernia CT assessment and improve multidisciplinary alignment in preoperative planning.
Keywords
Ventral hernia, computed tomography, standardized reporting, multi-criteria decision analysis, abdominal wall reconstruction
1. Introduction
Preoperative planning for abdominal wall reconstruction (AWR) is inherently complex, necessitated by the challenges of re-operative anatomy, the heterogeneity of available surgical approaches, and the expanding spectrum of prosthetic mesh materials. As such, precise and comprehensive preoperative assessment is essential for optimizing operative outcomes [1]. Unlike small, primary ventral hernias, complex or recurrent defects often cannot be reliably characterized through physical examination alone [2]. Consequently, preoperative imaging plays a pivotal role in procedural planning, with computed tomography (CT) becoming the primary imaging modality due to its ability to delineate hernia morphology and assess critical anatomical relationships [3].
Despite its established role in surgical planning, a persistent discrepancy exists between radiologic reporting and the informational requirements of the operating surgeon. While this discrepancy has not been extensively characterized in the literature, Kushner et al. have shown that radiologic reports frequently omit critical findings essential to preoperative decision-making [4]. In our clinical experience, radiology reports from our institution and referring centers commonly fail to include fundamental hernia-specific features. These include parameters, such as identification of the previously implanted mesh plane and characterization of mesh type, and more readily straightforward features, including defect dimensions, retained foreign bodies (e.g., fixation devices), and calculation of the Tanaka index. Given the central role of preoperative imaging in planning ventral hernia repair, one would expect the existence of standardized reporting criteria similar to established reporting protocols in other areas of surgery. However, to date, no consensus exists regarding which parameters should be routinely included in CT reports for ventral hernia evaluation [5].
This study aims to systematically evaluate the extent to which radiologists document ventral hernia–specific findings in CT scans obtained for preoperative assessment and to identify potential targets for standardization in radiologic reporting.
2. Methods
Following approval from the Cleveland Clinic Institutional Review Board, abdominopelvic CT reports were reviewed to assess the extent to which radiologists documented ventral hernia-specific features in adult patients scheduled for ventral hernia repair between January 2020 and December 2024 at the Cleveland Clinic Center for Abdominal Core Health (Cleveland, Ohio). To enable in-depth analysis of reporting practices in complex scenarios, we deliberately selected a subset of patients who underwent elective, open transversus abdominis release (TAR). Inclusion was restricted to cases in which CT scans were performed within six months before surgery and explicitly indicated ventral hernia repair as the intended procedure, ensuring that radiologists were aware of the surgical context. To further enrich the dataset with surgically meaningful findings, only patients with recurrent hernias previously managed with surgical intervention were included, allowing for assessment of whether prior repairs and implanted prosthetic materials were adequately documented in radiologic reports.
Eligible cases were identified through the abdominal core health quality collaborative (ACHQC). The ACHQC is a hernia-specific nationwide registry aimed at improving the quality of hernia care through patient-centered data collection, performance feedback to clinicians, and collaborative learning. Surgeons enter patient data prospectively in real-time during routine clinical care, including patient demographics, hernia characteristics, operative details, patient-reported outcomes (PROs), and postoperative follow-up information [6]. Once the relevant cohort was identified, corresponding CT reports were retrieved from the electronic medical record (EMR) using the Epic system. Only patients whose CT referrals met all predefined inclusion criteria were included in the final analysis.
To define the critical parameters for preoperative evaluation, we convened a panel of 12 AWR experts for structured discussion. The panel reached an expert consensus on a set of imaging features deemed essential for preoperative assessment and feasible to evaluate using CT. To supplement the expert input, a focused literature review was conducted to identify commonly cited CT-based parameters relevant to ventral hernia characterization and operative planning. The final list included 16 distinct radiologic features, each contributing unique and clinically significant information for surgical decision-making. These CT-based parameters were: defect width, defect length, number of defects, European Hernia Society classification for hernia location [7], Tanaka index [8], hernia content, presence of prior mesh, old mesh plane, central mesh fracture, mesh migration, presence of concurrent inguinal hernias, presence of concurrent parastomal or old stomal site hernias, abdominal wall anatomical abnormalities [9], rectus muscle measurements, patient habitus, and ongoing surgical site occurrences (SSOs). Further details regarding each CT-based parameter, including definitions, are provided in (Table 1).
TABLE 1: CT-based
parameters for planning AWR surgery.
|
# |
CT-based parameters |
Definitions |
AWR surgeons' rating (Mean ± SD) |
|
1 |
Defect width |
Measured as the transverse distance between the medial fascial edges at
the widest point of the hernia defect on axial CT view |
9.16 ± 2.02 |
|
2 |
Concurrent parastomal or old stomal site hernias |
Presence of a hernia adjacent to a stoma |
8.93 ± 1.91 |
|
3 |
Old mesh plane |
Determination of the anatomical layer in which the mesh is located
(onlay, sublay, and underlay) |
8.80 ± 2.25 |
|
4 |
Presence of prior mesh |
Documentation of whether previously placed prosthetic mesh is
visualized within the abdominal wall or peritoneal cavity |
8.61 ± 2.25 |
|
5 |
Concurrent inguinal hernias |
Identification of ipsilateral or contralateral inguinal hernias |
8.54 ± 2.18 |
|
6 |
Tanaka index |
Calculated as the ratio of hernia sac volume to total peritoneal volume |
8.45 ± 2.32 |
|
7 |
Hernia location (EHS classification) |
Anatomical location of the defect categorized according to the EHS
classification system |
8.36 ± 2.28 |
|
8 |
Abdominal wall anatomical abnormalities |
loss or scarring of native fascial planes—particularly the posterior
rectus sheath—often due to prior surgeries or infection. It is characterized
by the absence of reconstructable tissue layers, necessitating component
separation or myofascial release, and frequently requires biosynthetic or
staged repairs due to compromised tissue quality and contamination risk[9] |
8.17 ± 2.31 |
|
9 |
Number of defects |
Represents the total number of discrete fascial defects identified on
axial and coronal CT views |
8.05 ± 2.53 |
|
10 |
Rectus muscle measurements |
Quantitative assessment of rectus abdominis muscle width (from medial
to lateral) and depth (ventral to dorsal) in the widest part of the hernia as
seen in axial view |
7.98 ± 2.18 |
|
11 |
Patient habitus and fat distribution |
Evaluation of the predominant adipose tissue distribution, visceral
versus subcutaneous, typically inferred from skin-to-fascia and
fascia-to-bowel wall distances |
7.97 ± 2.54 |
|
12 |
Mesh migration |
Displacement of mesh from its intended anatomical location, either
partially or completely |
7.95 ± 2.36 |
|
13 |
Defect length |
Measured as the longitudinal distance from the most cephalad to the
most caudal extent of the fascial defect on a sagittal CT view |
7.66 ± 2.73 |
|
14 |
Central mesh fracture |
Evidence of structural discontinuity, segmentation, or tearing of a
previously placed mesh prosthesis |
7.64 ± 2.43 |
|
15 |
Hernia content |
Abdominal viscera in the hernia sac |
7.28 ± 2.98 |
|
16 |
Ongoing surgical site occurrences |
Presence of wound-related complications such as surgical site
infections (SSIs, per CDC definition), seroma, hematoma, abdominal wall
abscess or enterocutaneous fistula |
7.28 ± 2.47 |
To validate the clinical relevance of the identified CT-based parameters, we distributed an anonymous electronic survey to AWR specialists worldwide, including North America (United States and Canada), Western Europe (Germany, United Kingdom, France, and Spain), Australia, and Asia (India and Israel). The survey presented the list of 16 parameters and asked respondents to rate the clinical importance of each parameter on a scale from 1 to 10. Participants were also given the opportunity to suggest additional parameters they considered critical for preoperative planning.
To compare radiologic documentation with actual clinical characteristics, we extracted patient-specific hernia data from the ACHQC database and reviewed operative reports for all included patients. To assess the completeness of radiologic documentation of the 16 CT-based parameters, CT reports were evaluated independently by three reviewers using a standardized data abstraction chart. Quality assurance was maintained through calibration sessions, random audit checks, and intra-rater reliability assessments. Discrepancies were resolved by consensus, with a fourth reviewer consulted as needed. Reviewers were blinded to surgical characteristics. For meaningful assessment of the radiologic documentation, CT reports were then compared to actual clinical data. The 16 CT-based parameters were divided into two categories: eight generalizable parameters, defined as imaging features that can be evaluated independently of patient-specific surgical history. These included hernia defect width, defect length, number of defects, EHS classification for hernia location, Tanaka index, hernia content, rectus muscle measurements, and patient habitus. Documentation of these parameters was assessed using a binary scoring system, recording whether each feature was explicitly addressed in the radiologic report (yes/no). The second category included eight patient-specific parameters reflecting individualized clinical contexts and prior interventions. These encompassed the presence of previously implanted mesh, old mesh plane, central mesh fracture, mesh migration, presence of concurrent inguinal hernias, presence of concurrent parastomal or old stomal site hernias, abdominal wall anatomical abnormalities and ongoing SSOs. Documentation of these parameters was assessed by cross-referencing with operative findings and prospectively entered clinical data from ACHQC.
To establish a standardized set of CT-based parameters for abdominal wall reconstruction, we employed a systematic multi-criteria decision analysis (MCDA) approach integrating three distinct data sources. First, a panel of 12 AWR specialists conducted an expert audit, ranking 16 critical features through consensus discussion. Second, we surveyed 61 AWR surgeons globally who rated each feature's importance on a 10-point Likert scale. Finally, a systematic review of 40 studies assessed the frequency of feature documentation in the literature. Each data source was weighted according to its authority, relevance, and methodological strength using the analytic hierarchy process (AHP). The expert audit and systematic review received the highest weights due to their authoritative nature. Individual feature scores were calculated by combining normalized scores from each data source, multiplied by their respective weights. This comprehensive approach yielded a final prioritization ranking that balances clinical expertise, real-world practice patterns and literature evidence. Features were subsequently categorized into three tiers based on natural score clustering: Tier 1 (Essential, score ≥9.0), Tier 2 (Highly Recommended, score 6.0-9.0), and Tier 3 (Recommended, score <6.0). Detailed prioritization process can be seen in the (Supplementary Methods Section).
Statistical analysis included descriptive statistics for hernia characteristics and assessment of inter-observer agreement using Cohen's kappa coefficient. We used chi-square analysis to identify statistically significant differences between radiologists' reported intention to document features and actual documentation rates. All analyses used SPSS version 28.0 (IBM Corp., Armonk, NY), with statistical significance set at P < 0.05.
3. Results
Between January 2020 and December 2024, a total of 1,845 patients underwent elective TAR at the Cleveland Clinic Center for Abdominal Core Health. Of these, 834 patients met all predefined inclusion criteria (Figure 1). Baseline hernia and operative characteristics are summarized in (Table 2). Among the 834 patients with recurrent hernias, 337 (40.4%) had undergone one prior repair and 240 (28.8%) had undergone two; the remainder had experienced multiple recurrences. Previously implanted mesh was encountered intraoperatively in 71.1% of cases (593/834). Among these, mesh was identified in the retro-muscular plane in 821 patients (99.8%), with 516 (62.9%) also having preperitoneal mesh. Central mesh fracture was identified in 84 patients (10.1%), mesh migration in 77 patients (9.2%), and SSOs at the time of the index TAR procedure in 231 patients (27.7%). Concurrent stomas and inguinal hernias were each present in 34 patients (4.1%). Radiologic documentation of the eight generalizable CT-based parameters is presented in (Table 3). Defect width was reported in 272 reports (32.6%), defect length in 182 (21.8%), and number of defects in 262 (31.4%). Hernia location according to the EHS classification was reported in 222 cases (26.6%). The Tanaka index was documented in 108 reports (12.9%), and hernia content in 445 reports (53.4%). Rectus muscle measurements were documented in 3 cases (0.4%), and patient habitus in 2 cases (0.2%).
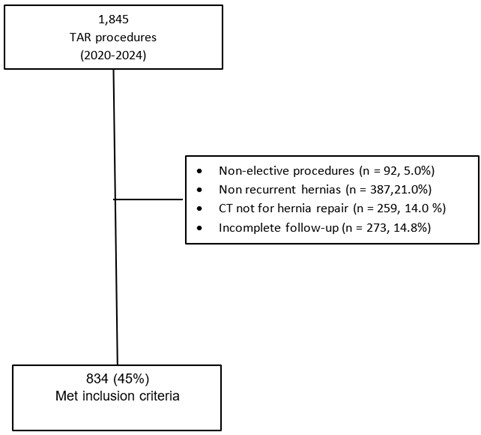
TABLE 2: Hernia and
operative characteristics.
|
Characteristic |
N = 834 |
|
|
Hernia etiology |
||
|
Primary |
0 (0%) |
|
|
Incisional |
834 (100%) |
|
|
Hernia type |
|
|
|
Medline |
762(91.4%) |
|
|
Flank hernia |
38(4.5%) |
|
|
Parastomal hernia |
34 (4.1%) |
|
|
Number of prior hernia repairs |
||
|
One prior hernia repair |
337 (40.4%) |
|
|
Two prior hernia repairs |
240 (28.8%) |
|
|
Three prior hernia repairs |
115 (13.8%) |
|
|
Four prior hernia repairs |
54 (6.5%) |
|
|
Five prior hernia repairs |
88 (10.6%) |
|
|
Concurrent inguinal hernias |
34 (4.1%) |
|
|
Prior mesh found in the current repair |
593 (71.1%) |
|
|
Prior mesh plane |
||
|
Onlay |
177 (30%) |
|
|
Sublay |
333 (56%) |
|
|
Underlay |
83 (14%) |
|
|
Hernia length, cm (mean ± SD) |
24.0 ± 6.2 |
|
|
Hernia width, cm (mean ± SD) |
17.0 ± 6.8 |
|
|
Mesh-related complications |
392 (47.0%) |
|
|
Mesh fracture in previous repair |
84 (10.1%) |
|
|
Mesh migration in previous repair |
77 (9.2%) |
|
|
SSO found during the current repair |
231 (27.7%) |
|
|
Surgical site infection |
46 (10.6%) |
|
|
Seroma |
25 (5.8%) |
|
|
Hematoma |
5 (1.2%) |
|
|
Fistula |
1 (0.2%) |
|
|
Others |
154 (18.5%) |
|
|
Intraoperative complication |
45(5.4%) |
|
|
Bowel injury |
16(1.9%) |
|
|
Operative time>2h |
733(87.9%) |
|
TABLE 3: Radiological
documentation of eight generalizable CT-based parameters.
|
Generalizable CT-based parameters |
Marked in the CT report (%) |
|
Defect width |
32.6% |
|
Defect length |
21.8% |
|
Number of defects |
31.4% |
|
Hernia location (EHS classification) |
26.6% |
|
Tanaka index |
12.9% |
|
Hernia content |
53.4% |
|
Rectus muscle measurements |
0.4% |
|
Patient habitus and fat distribution |
0.2% |
Documentation of the eight patient-specific CT-based parameters is shown in (Table 4). Among the 593 patients (71.1%) in whom previously implanted mesh was confirmed intraoperatively, 215 cases (36.2%) included documentation of mesh presence in the CT report. The anatomical mesh plane was specified in 10 cases (1.7%). Central mesh fracture was found intraoperatively in 84 patients (10.1%), and reported radiologically in 87 cases, exceeding the number of confirmed intraoperative findings (103.6%). Similarly, mesh migration was identified in 77 patients (9.2%) and reported in 86 CT interpretations (111.7%). Concurrent inguinal hernias, parastomal or old stomal site hernias were each identified intraoperatively in 34 patients (4.1%). However, CT reports documented inguinal hernias in 82 cases (241.2%) and parastomal hernias in 80 cases (235.3%). Abdominal wall anatomical abnormalities were identified intraoperatively in 250 patients (30.0%) and reported in 180 cases (72%). SSOs were present in 231 patients (27.7%) and were documented radiologically in 51 cases (22.1%).
TABLE 4: Radiological
documentation of eight patient-specific CT-based parameters.
|
specific CT-based parameters |
Occurrence (%) |
Marked in the CT report |
Marked in the CT report (% of Occurrence) |
|
Presence of prior mesh |
593 (71.1%) |
215 (25.8%) |
215 (36.2%) |
|
Old mesh plane |
593 (67.3%) |
10 (1.2%) |
10 (1.7%) |
|
Central mesh fracture |
84 (10.1%) |
87 (10.4%) |
87 (103.6%) |
|
Mesh migration |
77 (9.2%) |
86 (10.3%) |
86 (111.7%) |
|
Concurrent inguinal hernias |
34 (4.1%) |
82 (9.8%) |
82 (241.2%) |
|
Concurrent parastomal or old
stomal site hernias |
34 (4.1%) |
80 (9.6%) |
80 (235.3%) |
|
Abdominal wall anatomical abnormalities |
250 (30%) |
180 (21.6%) |
180 (72%) |
|
Ongoing surgical site occurrences |
231 (27.7%) |
51 (6.1%) |
51 (22.1%) |
A total of 61 AWR specialist surgeons completed the survey evaluating the perceived clinical relevance of the 16 defined CT-based parameters in the context of preoperative ventral hernia assessment. All parameters were considered clinically meaningful, each receiving a mean rating above 7 on a 10-point Likert scale. Defect width was rated highest (mean 9.16 ± 2.02), followed by presence of parastomal or old stomal site hernia (8.93 ± 1.91), anatomical mesh plane (8.80 ± 2.25), presence of prior mesh (8.61 ± 2.25), concurrent inguinal hernia (8.54 ± 2.18), Tanaka index (8.45 ± 2.32), EHS classification of hernia location (8.36 ± 2.28), and abdominal wall anatomical abnormalities (8.17 ± 2.31). The full ranking of all 16 parameters is shown in (Table 1).
Multi-criteria decision analysis outcomes are shown in (Table 5). The systematic application of MCDA with AHP weighting yielded a comprehensive ranking of all 16 CT-based parameters that produced final scores that ranged from 14.51 (defect width) to 3.58 (patient habitus and fat distribution). Tier 1 (score ≥9) included defect width, presence of prior mesh, old mesh plane, Tanaka index, hernia location and concurrent inguinal, parastomal and old stomal site hernias. Tier 2 (score 6.0-9.0) included anatomical abnormalities, rectus measurements and ongoing SSO. Tier 3 (score <6.0) included the remaining six features. The top 7 parameters demonstrated consistent high ratings across all evaluative domains and achieved a MCDA score of 9 or higher. Notably, defect width score reflects its importance across all data sources: highest surgeon audit ranking (16/16), highest survey rating (9.16/10), most cited in literature. (Figure 2).
TABLE 5: Final prioritization of CT-based parameters using multi-criteria decision
analysis.
|
Rank |
Parameter |
Final Score |
Tier |
Recommendation |
|
1 |
Defect width |
14.51 |
1 |
Essential |
|
2 |
Presence of prior mesh |
13.21 |
1 |
Essential |
|
3 |
Old mesh plane |
12.15 |
1 |
Essential |
|
4 |
Tanaka index |
12.09 |
1 |
Essential |
|
5 |
Hernia location (EHS) |
11.13 |
1 |
Essential |
|
6 |
Concurrent parastomal or old
stomal site hernias |
10.00 |
1 |
Essential |
|
7 |
Concurrent inguinal hernias |
9.52 |
1 |
Essential |
|
8 |
Abdominal wall anatomical abnormalities |
8.73 |
2 |
Highly Recommended |
|
9 |
Rectus muscle measurements |
7.58 |
2 |
Highly Recommended |
|
10 |
Ongoing surgical site occurrences |
6.94 |
2 |
Highly Recommended |
|
11 |
Central mesh fracture |
5.68 |
3 |
Recommended |
|
12 |
Mesh migration |
5.63 |
3 |
Recommended |
|
13 |
Defect length |
4.61 |
3 |
Recommended |
|
14 |
Hernia content |
4.60 |
3 |
Recommended |
|
15 |
Patient habitus and fat distribution |
4.28 |
3 |
Recommended |
|
16 |
Number of defects |
3.58 |
3 |
Recommended |

4. Discussion
Our findings reveal substantial discrepancies between the informational requirements of surgeons performing ventral hernia repair and the current standards of CT reporting. Overall documentation completeness was limited, with a median of 34.4%. Among the eight generalizable CT-based parameters, the median reporting rate was only 24.2% (range: 0.2%-53.4%). Patient-specific parameters demonstrated a higher median reporting rate of 87.8% when the condition was present (range: 1.7%-241.2%). While some parameters were reliably captured, such as central mesh fracture and mesh migration, which were documented at rates exceeding intraoperative detection (103.6% and 111.7%, respectively)—the general performance of CT reporting fell short of surgical expectations. Notably, defect width- despite being rated as the most critical parameter- was documented in just 32.6% of reports. Parameters such as rectus muscle measurements and patient habitus, both critical for operative planning and mesh selection, were documented in only 0.4% and 0.2% of reports, respectively. In contrast to these generalizable parameters, patient-specific findings showed variable documentation rates. While mesh presence was documented in only 36.2% of cases where mesh was found intraoperatively, and the old mesh plane was specified in just 1.7% of cases, other patient-specific parameters like concurrent hernias were over-reported relative to their actual occurrence.
To our knowledge, this study represents the largest systematic evaluation of CT reporting practices for ventral hernia repair, encompassing 834 consecutive cases. Despite the critical importance of accurate and complete radiologic documentation in guiding preoperative strategy, most surgeons continue to rely on direct image interpretation rather than radiology reports [10]. This reliance reflects a significant gap in current practice, particularly in healthcare settings where surgeons may not have routine or immediate access to imaging platforms. In such contexts, comprehensive and standardized radiologic reporting is essential to ensure accurate preoperative assessment, facilitate operative planning, and support informed patient counselling.
Existing literature suggests that the deficiencies identified in our study reflect systemic rather than institution-specific issues in radiologic reporting practices. Halligan et al. [1, 11] conducted a multicenter study across five European specialized hernia centers and found that only 12% of centers had established protocols for hernia-specific CT reporting, with 78% of surgeons routinely re-reviewing images due to inadequate radiologic documentation. Our deliberate inclusion of patients with recurrent ventral hernias undergoing open TAR permitted a focused evaluation in anatomically complex cases, where comprehensive and precise preoperative imaging is essential. Given the limited literature on this topic, the persistence of documentation gaps in our cohort—despite being drawn from a high-volume hernia center where radiologists routinely interpret imaging studies involving complex abdominal wall defects—underscores the broader relevance of our findings. These results suggest that the need for standardized CT reporting criteria in ventral hernia care is not confined to isolated practices but rather represents a widespread challenge across diverse clinical settings.
The reporting deficiencies identified in this study appear to exceed those documented in other surgical subspecialties. Nörenberg et al. [12] reported that despite standardized templates for rectal MRI, critical features were reported in only 38-52% of cases. Brook et al. [13] demonstrated similar gaps in pancreatic adenocarcinoma reporting, with resectability features documented in 45-51% of reports. In comparison, our findings demonstrate discrepancies of up to 70% between surgical findings and radiologic documentation, underscoring a more pronounced deficiency in the context of abdominal wall imaging. These findings suggest that the need for standardization in CT reporting may be particularly urgent in the evaluation of ventral hernias. The importance of standardized operative documentation has been similarly emphasized [14], and multidisciplinary team approaches have shown improved outcomes [15].
Several theoretical explanations may account for the observed discrepancies between the informational needs of surgeons and current CT reporting practices. One possibility is that radiologists may lack familiarity with certain hernia-specific parameters that are considered critical by AWR surgeons. Alternatively, some features may not be reliably discernible on CT imaging. Another contributing factor may be a limited awareness among radiologists of the clinical relevance of specific parameters that are considered essential by surgeons during operative planning. As this study was not designed to investigate the causes of these discrepancies, we are unable to definitively determine their origin. Nonetheless, to meaningfully improve reporting standards, we believe an important initial step would be the establishment of a clearly defined set of parameters considered essential by AWR surgeons for preoperative planning. The ultimate goal is to ensure that, in the event a patient presents with only a radiology report without access to the original CT images, surgical decision-making can still proceed without necessitating repeat imaging.
To prioritize the essential CT parameters out of the sixteen CT based parameters we applied a MCDA using the analytic hierarchy process. Parameters with an MCDA score of ten or higher, including those in Tier 1 and the upper median of Tier 2, were identified as most critical, supporting their prioritization for standardized CT reporting. These parameters include defect width, presence of prior mesh, old mesh plane, Tanaka index, hernia location (EHS classification), concurrent inguinal hernia and concurrent parastomal or old stomal site hernia. In response, we modestly propose a pragmatic framework for abdominal wall imaging based on the mnemonic "STAMP-C":
• Size-defect width.
• Tanaka index.
• Anatomical location-hernia location (EHS).
• Mesh-presence of prior mesh.
• Plane-old mesh plane.
• Concurrent hernia - Concurrent inguinal and parastomal or old stomal site hernia.
This study is subject to several limitations. First, although this single-center design may limit generalizability, several factors support the broader applicability of our findings. Our global surgeon survey included 61 AWR specialists from North America, Western Europe, Australia, and Asia, providing international perspective on parameter prioritization. Additionally, the consistency of our findings with published literature from multiple institutions suggests these reporting deficiencies are systemic rather than institution-specific. To further address generalizability, we are establishing an international consortium including centers from the United States, Europe, and Asia-Pacific regions for the prospective validation study. This multi-center approach will ensure the STAMP-C framework's applicability across diverse healthcare systems, practice patterns, and patient populations. Second, the use of operative documentation as the reference standard introduces the possibility of incomplete or inconsistent reporting. To mitigate this, we cross-referenced operative notes with data from the ACHQC registry to enhance accuracy and completeness. Importantly, some of the observed reporting deficiencies were not confined to surgeon-dependent findings. Generalizable CT-based parameters, those discernible independent of operative documentation, demonstrated similarly low rates of reporting. Our use of a binary scoring approach, while effective for evaluating the presence or absence of documentation, did not assess the accuracy or qualitative adequacy of the reported findings. The proposed set of essential parameters for preoperative planning was developed through expert consensus, an AWR surgeon survey, and informed by relevant literature; however, it has not been prospectively validated in a clinical trial. As such, its adoption should be approached with caution and adapted to local practice as appropriate. Additionally, while our expert panel comprised experienced AWR surgeons, the absence of radiologist representation in the consensus process represents a limitation. Future iterations of this framework should incorporate radiologist perspectives to enhance interdisciplinary collaboration and ensure practical feasibility of implementation from the imaging perspective. Finally, this study focused exclusively on patients with complex, recurrent hernias requiring TAR procedures. While this selection was intentional to emphasize the need for detailed radiologic assessment in anatomically challenging cases, it may not fully reflect routine reporting practices for less complex presentations.
While this study provides a comprehensive framework for standardizing CT reporting, prospective validation represents a critical next step. We are currently developing a multi-institutional trial protocol to implement the STAMP-C framework across diverse practice settings. This prospective study will assess: i) improvements in surgical planning efficiency through time-to-decision metrics, ii) reduction in repeat imaging requests, iii) inter-rater reliability among radiologists using the framework, and iv) correlation with patient outcomes including operative time, complication rates, and recurrence. We anticipate launching this validation study across five centers within the next six months, with results available to guide broader implementation. For the prospective validation phase, we have assembled a multidisciplinary steering committee that includes both AWR surgeons and abdominal radiologists. This collaborative approach will ensure that the STAMP-C framework is both clinically relevant and practically implementable within radiology workflows. We believe this surgeon-radiologist partnership will be crucial for successful adoption and sustainability of standardized reporting practices.
5. Conclusion
This study demonstrates a substantial gap between the radiologic documentation provided in preoperative CT reports and the informational needs of surgeons performing abdominal wall reconstruction. By applying a structured methodology to prioritize essential radiologic features, we offer a core set of parameters deemed most critical for surgical planning. The proposed "STAMP-C" framework offers a practical, qualitative template that aligns with radiologic reporting norms while addressing surgical priorities. We encourage other institutions and professional societies to assess, refine, and validate this proposed framework in diverse clinical settings to promote broader standardization and enhance multidisciplinary collaboration in the care of patients with complex ventral hernias.
Acknowledgements
The authors thank the surgeons and radiologists who participated in our survey, the radiology department staff for their assistance in accessing imaging records, and the research staff who contributed to data collection and analysis.
Disclosures
The authors declare that they have no financial or personal relationships with other people or organizations that could inappropriately influence (bias) their work. No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this manuscript. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
None.
Conflicts of Interest
None.
Abbreviations
AWR: Abdominal Wall Reconstruction
CT: Computed Tomography
TAR: Transversus Abdominis Release
EHS: European Hernia Society
SSI: Surgical Site Infection
ASA: American Society of Anesthesiologists
REFERENCES
1.
Steve Halligan, Sam G Parker, Andrew A Plumb, et al.
“Imaging complex ventral hernias, their surgical repair, and their
complications.” Eur Radiol, vol. 28, no. 8, pp. 3560-3569, 2018. View
at: Publisher Site | PubMed
2.
Julie L Holihan, Burzeen Karanjawala, Annie Ko, et al.
“Use of Computed Tomography in Diagnosing Ventral Hernia Recurrence.” JAMA
Surg, vol. 151, no. 1, pp. 7-13, 2016. View at: Publisher
Site | PubMed
3.
Laurel J Blair, Samuel W Ross, Ciara R Huntington, et
al. “Computed tomographic measurements predict component separation in ventral
hernia repair.” J Surg Res, vol. 199, no. 2, pp. 420-427, 2015. View at:
Publisher
Site | PubMed
4.
Bradley Kushner, Carter Starnes, Maggie Sehnert, et
al. “Identifying critical computed tomography (CT) imaging findings for the
preoperative planning of ventral hernia repairs.” Hernia, vol. 25, no.
4, pp. 963-969, 2021. View at: Publisher
Site | PubMed
5.
Jignesh A Gandhi, Pravin H Shinde, Amay M Banker, et
al. “Computed tomography for ventral hernia: Need for a standardised reporting
format.” J Minim Access Surg, vol. 19, no. 2, pp. 175-177, 2023. View
at: Publisher Site | PubMed
6.
Nir Messer, Megan S Melland, Benjamin T Miller, et al.
“Evaluating the impact of lifting mandatory smoking cessation prior to elective
abdominal wall reconstruction. A single-center experience.” Am J Surg,
vol. 229, pp. 52-56, 2024. View at: Publisher
Site | PubMed
7.
F E Muysoms, M Miserez, F Berrevoet, et al.
“Classification of primary and incisional abdominal wall hernias.” Hernia,
vol. 13, no. 4, pp. 407-414, 2009. View at: Publisher
Site | PubMed
8.
E Y Tanaka, J H Yoo, A J Rodrigues Jr, et al. “A
computerized tomography scan method for calculating the hernia sac and
abdominal cavity volume in complex large incisional hernia with loss of
domain.” Hernia, vol. 14, no. 1, pp. 63-69, 2010. View at: Publisher
Site | PubMed
9.
Michael J Rosen, Joel J Bauer, Marco Harmaty, et al.
“Multicenter, Prospective, Longitudinal Study of the Recurrence, Surgical Site
Infection, and Quality of Life After Contaminated Ventral Hernia Repair Using
Biosynthetic Absorbable Mesh: The COBRA Study.” Ann Surg, vol. 265, no.
1, pp. 205-211, 2017. View at: Publisher
Site | PubMed
10. Jonathan L
Mezrich “Legal perspective: surgeons and imaging--are self-reads a mistake?” Am
J Surg, vol. 204, no. 4, pp. 545-547, 2012. View at: Publisher
Site | PubMed
11. Steve Halligan,
Sam G Parker, Andrew A O Plumb, et al. “Use of imaging for pre- and
post-operative characterisation of ventral hernia: systematic review.” Br J
Radiol, vol. 91, no. 1089, pp. 20170954, 2018. View at: Publisher Site | PubMed
12. Dominik
Nörenberg, Wieland H Sommer, Wolfgang Thasler, et al. “Structured Reporting of
Rectal Magnetic Resonance Imaging in Suspected Primary Rectal Cancer.” Invest
Radiol, vol. 52, no. 4, pp. 232-239, 2017. View at: Publisher
Site | PubMed
13. Olga R Brook,
Alexander Brook, Charles M Vollmer, et al. “Structured Reporting of Multiphasic
CT for Pancreatic Cancer: Potential Effect on Staging and Surgical Planning.” Radiology,
vol. 274, no. 2, pp. 464-472, 2015. View at: Publisher
Site | PubMed
14. Naila H
Dhanani, Nicole B Lyons, Celia M Divino, et al. “Expert Consensus for Key
Features of Operative Reports of Ventral Hernia.” J Am Coll Surg, vol.
236, no. 2, pp. 235-240, 2023. View at: Publisher
Site | PubMed
15. S G Parker, H Blake, S Zhao, et al. “An established abdominal wall multidisciplinary team improves patient care and aids surgical decision making with complex ventral hernia patients.” Ann R Coll Surg Engl, vol. 106, no. 1, pp. 29-35, 2024. View at: Publisher Site | PubMed
